Z E M C H 2 0 1 2 I n t e r n a t i o n a l C o n f e r e n c e
670
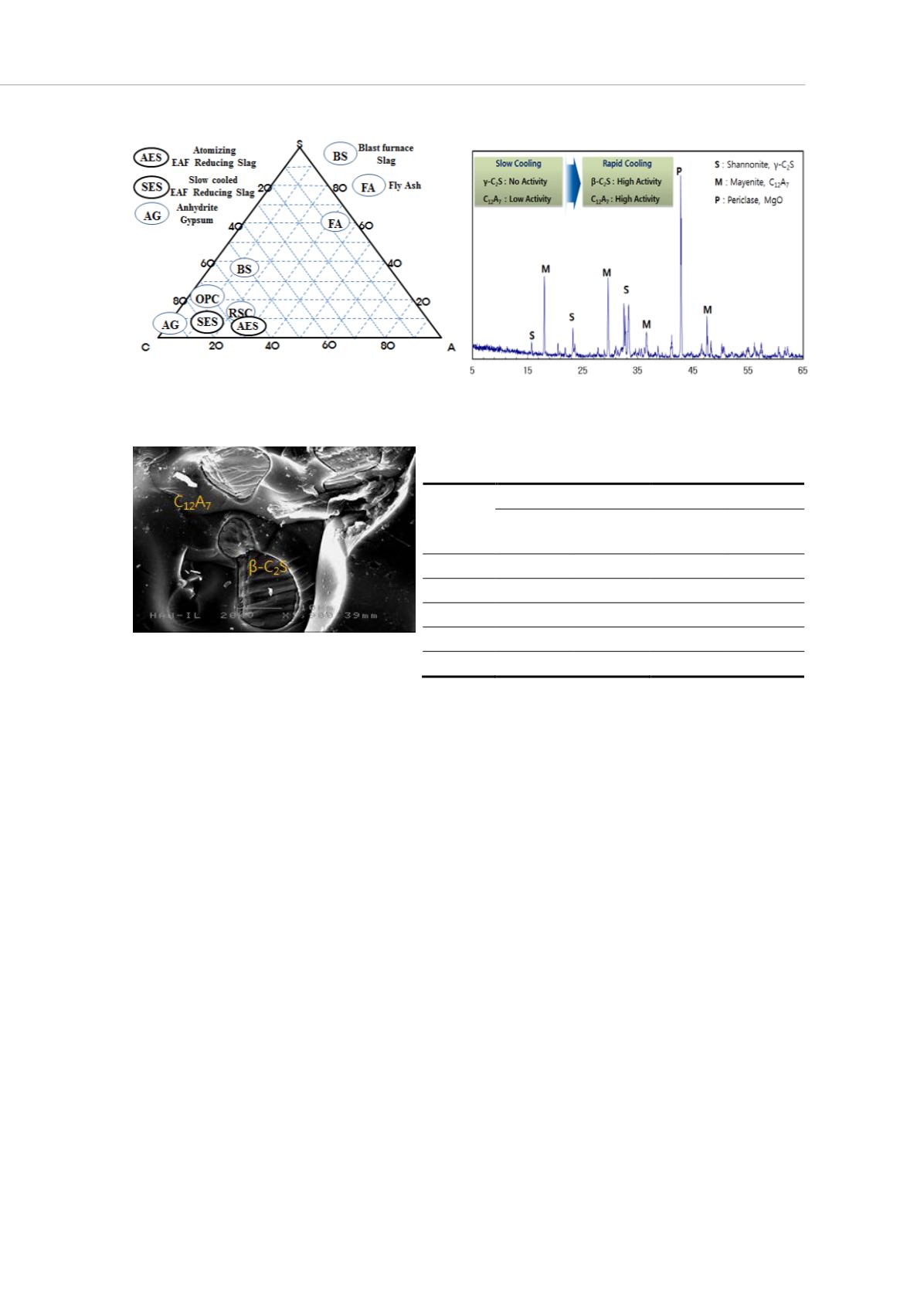
Figure 1 : Phase diagram of Atomizing EAF
Reducing Slag powder
Figure 2 : X-ray diffraction patterns of atomizing
EAF Reducing Slag powder
Figure 3 : SEM Image of the
EAFRS(X1,900)
Table 3 : EDS results of EAFRS
Eleme
nt
C
12
A
7
β-C
2
S
Weight
%
Atomic
%
Weight
%
Atomic
%
O
46.33
64.36
35.70
55.66
Al
17.91
14.75
-
-
Si
4.50
3.56
16.30
14.48
Ca
31.27
17.34
47.99
29.86
Total
100%
100%
3.2 Setting time
Figure 4 shows the setting time of RC EAFRS powder and RSC. Since RC EAFRS
powder immediately reacts with water and quick set so that it cannot be worked when it
contacts water, retarder for RSC was used same in the RC EAFRS powder too. Setting
time of RSC was measured using 0.5% (compared to binder) recommended retarder use.
As shown in Figure 4 showing the retarder use of RC EAFRS, if retarder 0.5% is used
same as RSC, it shows faster initial setting time and final setting time than RSC.
Citric acid used as retarder consumes Ca(OH)
2
created by Ca chloride at the initial
phase of hydration, lowers the density of Ca ion in the liquid and raises the ratio of
aluminum hydroxide gel containing fluorine in the hydrate. Since this gel has big volume,
it restricts the generation and growth of ettringite by covering the unhydrated C
11
A
7
CaF
2
particle surface.
2)
As the Ca ion in liquid is consumed by reaction and lowers to some level, it is rapidly
dissolved in the aluminum hydroxide liquid containing fluorine, and hydration reaction of
C
11
A
7
CaF
2
actively occurs again. This is why time difference of the initial setting time and
final setting time difference between RC EAFRS powder and regulated set cement is not
that big. Since the setting time can be adjusted with amount of citric acid addition, it is
extremely effective as the retarder to obtain workability of RC EAFRS powder.


